Orthopedic devices that contain canted coil springs can be cleaned by methods that meet FDA requirements. Two such validated methods are discussed below.
Steve Twork, Global Market Manager, Medical Device
According to the Food and Drug Administration (FDA), inadequate reprocessing of medical devices between patients can result in the retention of blood, tissue and other biological debris or soil in certain types of reusable medical devices. Such debris, says the agency, can allow microbes to survive the disinfection or sterilization process, which could then lead to healthcare-associated infections (HAIs) and other adverse patient outcomes.
 The FDA also notes that reducing the risk of exposure to improperly reprocessed medical devices is a shared responsibility of FDA, healthcare facilities and, of course, manufacturers, who are responsible for providing “adequate instructions that are user friendly and proven to work.”1
The FDA also notes that reducing the risk of exposure to improperly reprocessed medical devices is a shared responsibility of FDA, healthcare facilities and, of course, manufacturers, who are responsible for providing “adequate instructions that are user friendly and proven to work.”1
The Bal Spring™ canted coil spring is integrated into a variety of orthopedic and surgical tools to perform latching, locking and holding functions. As a component in these devices, it is essential that the spring is easy to clean according to medical device industry standards and FDA regulations. This article presents two methods suitable for cleaning orthopedic surgical instruments that contain canted coil springs.
In the first method, Bal Seal Engineering, Inc. engaged a third-party test house to validate its recommended cleaning process. The tests were conducted according to industry standards and FDA requirements. The second method presented is the process that Symmetry Medical recommends for cleaning and reprocessing its reusable orthopedic products, some of which contain Bal Spring™ canted coil springs. An overview of this cleaning method is discussed. For both methods, the protocols must be verified and validated before use.
Bal Seal Engineering’s Cleaning Method
The goal of engaging a third party to test Bal Seal Engineering’s protocol was to demonstrate that devices containing Bal Spring™ canted coil springs can be cleaned (i.e., organisms and other soil components can be removed) using the manual cleaning method as described below. The test, which was conducted under worst-case conditions, validated that once a device containing a canted coil spring is used, it can be properly cleaned by either the manual or automated cleaning method recommended by Bal Seal Engineering.
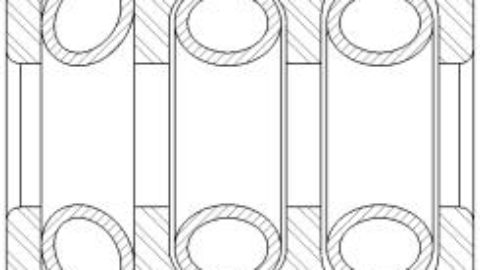
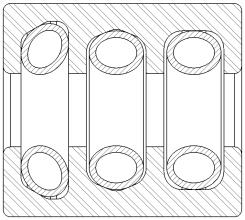 For testing, the company selected common orthopedic devices consisting of a single housing with multiple groove configurations with a Bal Spring™ canted coil spring (BSE 100 series) in each groove (see Figure 1). The cleaning validation was performed in accordance with the AAMI TIR30:2011 guidance document, “A compendium of processes, materials, test methods, and acceptance criteria for cleaning reusable medical devices.”2
For testing, the company selected common orthopedic devices consisting of a single housing with multiple groove configurations with a Bal Spring™ canted coil spring (BSE 100 series) in each groove (see Figure 1). The cleaning validation was performed in accordance with the AAMI TIR30:2011 guidance document, “A compendium of processes, materials, test methods, and acceptance criteria for cleaning reusable medical devices.”2
Defibrinated blood soil was used to contaminate the devices. The residuals tested included hemoglobin and protein. For this manual cleaning method, the devices were fully immersed in prepared blood soil and were allowed to remain in contact with the blood soil for a minimum of 15 minutes. After the 15-minute contact time, the devices were removed from the blood soil and allowed to dry uncovered at room temperature for a minimum of one hour.
Each device was rinsed using a water jet pistol filled with cool tap water at approximately 40 psi. During rinsing, a soft bristle brush and pipe cleaner were used to thoroughly scrub the devices to ensure removal of gross contamination. Each device was then immersed in enzymatic detergent such as Enzol at 1 oz/gal using lukewarm tap water and allowed to soak for a minimum of one minute. After soaking, each device was brushed with an appropriately sized pipe cleaner beneath the surface of the prepared detergent for a minimum of one minute. It was then rinsed with the water jet pistol to remove the residue. After ensuring that the devices were free of debris and detergent residue, they were dried with a clean, soft, lint-free cloth and/or a filter pressurized at ≤20 psi. Flushing and full-immersion manual shaking was used to extract fluid, which was tested to determine the levels of hemoglobin and protein on each device. Sterile water was used for irrigation.
Two tests—the hemoglobin test and the Micro BCA protein test—were conducted to determine whether the devices were sufficiently cleaned. For the hemoglobin test, the limit of detection (LOD) was calculated to be 0.16 µg/mL. This value was used in the calculations for test devices with results below the detection limit. The limit of quantitation (LOQ) for the method was calculated to be 0.5 µg/mL. In cases in which the µg/mL value was between the LOD and LOQ, the calculated results were estimated. The hemoglobin concentrations shown in Table I represent the average of the three replicates.
| Article Number | µg/mL | µg/article | µg/cm2 |
|---|---|---|---|
| 1 | < 0.16 | < 3.2 | < 0.37 |
| 2 | 0.18 | 3.6 | 0.42 |
| 3 | 0.34 | 6.8 | 0.79 |
| Negative Article | 1.3 | 25 | 2.9 |
| Positive Article | 2.4 | 4,800 | 560 |
Table I. Hemoglobin test results.
For the Micro BCA protein test, the LOD for the assay was calculated to be 1.1 µg/mL. This value was used in the calculations for the test devices for which the absorbance was below the detection limit. The LOQ for the assay was calculated to be 2 µg/mL. In cases in which the µg/mL value was between the LOD and the LOQ, the calculated results were estimated (see Table II).
| Article Number | µg/mL | µg/article | µg/cm² |
|---|---|---|---|
| 1 | < 1.1 | < 22 | < 2.6 |
| 2 | < 1.1 | < 22 | < 2.6 |
| 3 | 2.3 | 46 | 5.3 |
| Negative Article | 1.4 | 28 | 3.2 |
| Positive Article | 590 | 12,000 | 1,400 |
Table II. Micro BCA™ protein test results.
In order for the test analysis to be considered valid, acceptance criteria had to be met or sufficiently justified. The acceptance criteria were defined as a positive titer on the device being tested to produce a desired log10 reduction for the processed devices. However, because soil and organisms were sometimes incompatible with all materials (for example, the soil or organism did not adhere to materials or it was susceptible to antimicrobial agents), some devices had to be recontaminated with higher titer concentrations or via special methods.
Following the cleaning procedure, no soil was seen on the processed test devices under normal lighting conditions during visual inspection. The procedure sufficiently cleaned the tested devices to remove organisms and other soil components.
The Symmetry Medical Cleaning Method
Symmetry Medical, a maker of orthopedic implants and surgical instruments, has issued its own cleaning instructions for reusable surgical instruments, which includes devices that may contain one or more Bal Spring™ canted coil springs. According to the document, titled “Recommended Care, Cleaning, and Sterilization for Reusable Instruments, Instructions for Use,” all Symmetry Medical instruments may be safely and effectively reprocessed using the manual or combination manual/automated cleaning instructions provided in the document “unless otherwise noted in instructions accompanying a specific instrument.”3
The company’s instructions are written in accordance with ISO 17664, “Sterilization of medical devices — Information to be provided by the manufacturer for the processing of resterilizable medical devices.”4
This standard sets out the specific information medical device manufacturers must provide “on the processing of medical devices claimed to be resterilizable, and medical devices intended to be sterilized by the processor.” It defines the types of information required to ensure that a medical device can be processed safely so that it will continue to meet its performance specification.
According to Symmetry, its reprocessing instructions have been validated as being capable of preparing reusable Symmetry instruments for use. The document provides specific warnings, precautions and limitations on processing along with the thorough cleaning instructions. While detailing those caveats is beyond the scope of this article, it is imperative that users read and understand them prior to implementing this cleaning process. All of the company’s protocols must be verified before being put into practice.
In this method, excess biologic soil is removed from the instruments with a disposable wipe. Devices are placed in a container of distilled water or covered with damp towels. The company notes that soaking devices in proteolytic enzyme solution facilitates cleaning—especially in instruments with complex features such as lumens, mating surfaces, blind holes and cannulas. Instruments that cannot be soaked or maintained damp should be cleaned within 30 minutes of use to minimize the potential for drying prior to cleaning. The Symmetry method requires that multicomponent instruments be disassembled before cleaning.
Symmetry’s guide outlines both a manual cleaning process and a combination manual/automated process. The manual process is broken down into nine steps while the combined process contains seven. For this article, the manual process is discussed. The overview below summarizes the nine steps presented in Symmetry’s instructions.
For this method, instruments are completely submerged in a proteolytic enzyme solution that is prepared according to the manufacturer’s instructions. The instruments are then gently shaken to remove trapped bubbles. Instruments with hinges or moving parts are actuated to ensure contact of the solution with all surfaces. Lumens, blind holes and cannulations should be flushed with a syringe to remove bubbles and to ensure contact of the solution with all instrument surfaces.
Instruments are soaked for a minimum of 10 minutes, and surfaces are scrubbed using a soft nylon-bristled brush until all visible soil has been removed. Symmetry specifically notes that particular attention should be given to crevices, roughened surfaces, cutting features, hinged joints, sharp edges, box locks and areas with small components or springs. The company recommends inserting a long narrow nylon bristle brush/pipe cleaner into the lumen, blind hole or cannula with a twisting motion while pushing in and out multiple times to remove soil.
While the device is immersed, all moving parts of the instruments should be actuated to ensure that the cleaning solution is completely exposed to all surfaces. Scrubbing devices below the surface of the enzyme solution minimizes the potential of aerosolizing contaminated solution.
At this stage, the instruments are removed from the enzyme solution, rinsed and then actuated in tap water for a minimum of one minute. Symmetry recommends thoroughly and aggressively flushing lumens, holes, cannulas and other difficult to access areas. This step would apply equally to canted coil springs.
Instruments are then completely submerged in the cleaning solution—an ultrasonic cleaning bath with detergent and de-gas—and gently shaken again to remove any trapped bubbles. The lumens, blind holes and cannulations are to be flushed with a syringe to remove bubbles and ensure contact of the solution with all instrument surfaces. A minimum of 10 minutes is suggested for sonic cleaning. Symmetry also recommends separating stainless steel instruments from other metal instruments during ultrasonic cleaning to avoid electrolysis. The instructions note that hinged instruments should be fully opened and that wire mesh baskets or trays designed for ultrasonic cleaners should be used. Regular monitoring with an ultrasonic activity detector, aluminum foil test, TOSI or SonoCheck is recommended.
After removing the instruments from the ultrasonic bath, they should be rinsed in purified water for a minimum of one minute or until there is no sign of residue detergent or biologic soil. The instructions specify actuating all movable and hinged parts while rinsing and thoroughly and aggressively flushing lumens, holes, cannulas and other complex areas to access, such as canted coil springs.
Like the validated Bal Seal Engineering method described earlier, Symmetry also specifies that all devices are to be thoroughly inspected for residual biologic soil or detergent. If contamination is still present, the cleaning process should be repeated. Symmetry requires that its devices be sterilized prior to use and provides specific sterilization instructions at the end of the manual.
Conclusion
Devices containing the Bal Spring™ canted coil spring can be cleaned with methods that meet FDA regulations and industry standards. Two validated methods have been discussed that follow industry standards to safely and effectively reprocess orthopedic surgical instruments. Bal Seal Engineering, Inc. engaged a third-party test house to validate the cleaning process it recommends for orthopedic products. The method required by Symmetry Medical has also been validated and is suitable for cleaning orthopedic products that contain Bal Spring™ canted coil springs.
References
1. “Reprocessing of Reusable Medical Devices,” http://www.fda.gov/medicaldevices/deviceregulationandGuidance/reprocessingofreusablemedicaldevices/default.htm, accessed April 29, 2014.
2. AAMI TIR30:2011 guidance document, “A compendium of processes, materials, test methods, and acceptance criteria for cleaning reusable medical devices,” Association for the Advancement of Medical Instrumentation, August 10, 2011.
3. “Recommended Care, Cleaning, and Sterilization for Reusable Instruments, Instructions for Use,” Symmetry Medical, 7001-99 Rev. E.
4. ISO 17664, “Sterilization of medical devices — Information to be provided by the manufacturer for the processing of resterilizable medical devices,” International Organization for Standardization, 2004.
Steve Twork is the global market manager for medical devices at Bal Seal Engineering, Inc., a global provider of custom-engineered sealing, connecting, conducting and EMI shielding solutions for industry worldwide. The company’s products employ unique Bal Spring™ canted coil spring technology for enhanced equipment performance and reliability. For more information about Bal Seal Engineering products and services, e-mail sales@balseal.com or call 949-460-2100.